Precision Oncology Reimagined: Smart Nano carriers and AI-Guided Multi-Omics Strategies
Keywords:
AI-integrated nanomedicine; precision oncology; smart nanocarriers; multi-omics; hepatocellular carcinoma; breast cancer; renal cell carcinoma; glioblastoma; targeted therapy; theranosticsAbstract
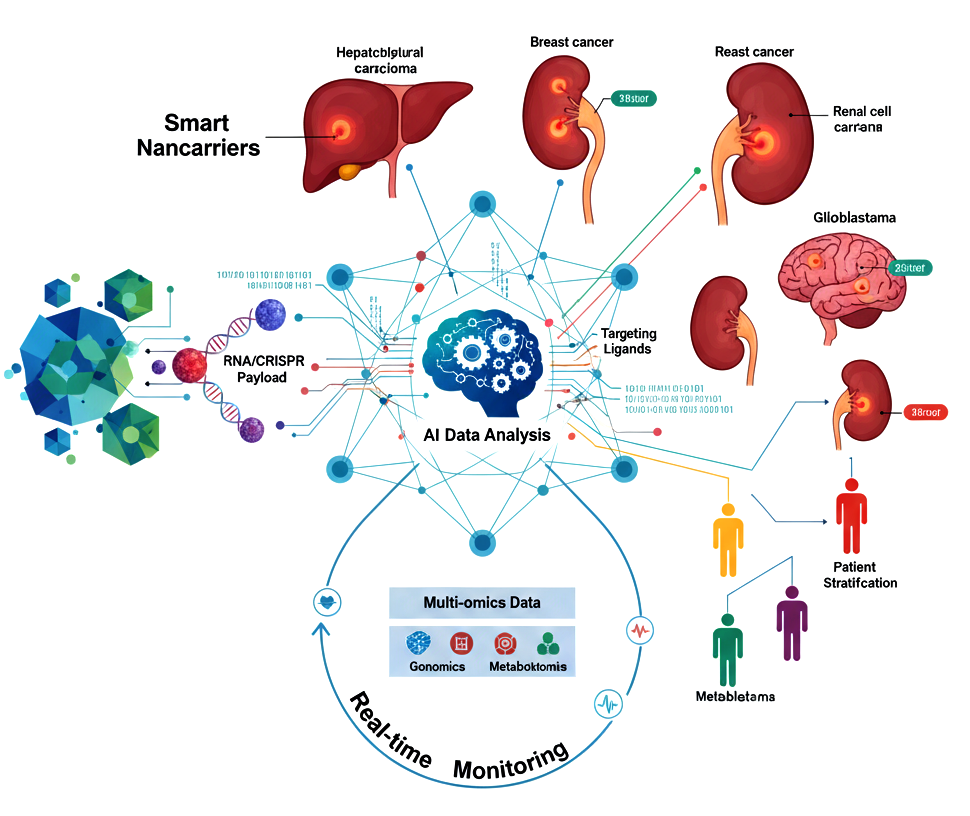
Background: Conventional cancer therapies are limited by systemic toxicity, poor specificity, and tumor heterogeneity. The convergence of smart nanocarriers, artificial intelligence (AI), and multi-omics profiling offers transformative potential in precision oncology. Objectives: This review summarizes advances in AI-integrated nanotherapeutics (2019–2024), focusing on liver, breast, renal, and neuro cancers, and evaluates their clinical translation, therapeutic efficacy, and future directions. Methods: Literature from 2019–2024 was systematically analyzed, emphasizing nanocarrier design, therapeutic payloads, AI-guided target selection, patient stratification, and clinical trials. Multi-omics data (genomics, transcriptomics, proteomics, metabolomics, and single-cell analyses) were reviewed for their role in guiding personalized therapy. Results: Smart nanocarriers lipid-based, polymeric, inorganic, biomimetic, and exosome-derived enable precise delivery of chemotherapeutics, RNA therapeutics, CRISPR-Cas systems, and immunomodulators. AI enhances nanoparticle design, predicts therapeutic response, and informs patient stratification. Multi-omics integration enables adaptive, individualized therapy, improving tumor targeting, overcoming resistance, and optimizing combinatorial approaches. Early-phase clinical trials demonstrate feasibility, safety, and preliminary efficacy across all four cancer types. Conclusions: AI-integrated, multi-omics-guided nanomedicine represents a paradigm shift in precision oncology, enabling personalized, adaptive, and multifunctional cancer therapies. Future research should focus on scalable manufacturing, safety validation, ethical deployment, and global clinical adoption.